Mosaic Aging: Why Your Organs Age at Different Rates, and Why It Matters
A growing body of work, capped by an ambitious 2026 framework, argues that the body does not age all at once. It ages in tiles. The pace at which the fastest-aging tile breaks down may be what determines how long you live.
Look at any seventy-year-old you know.
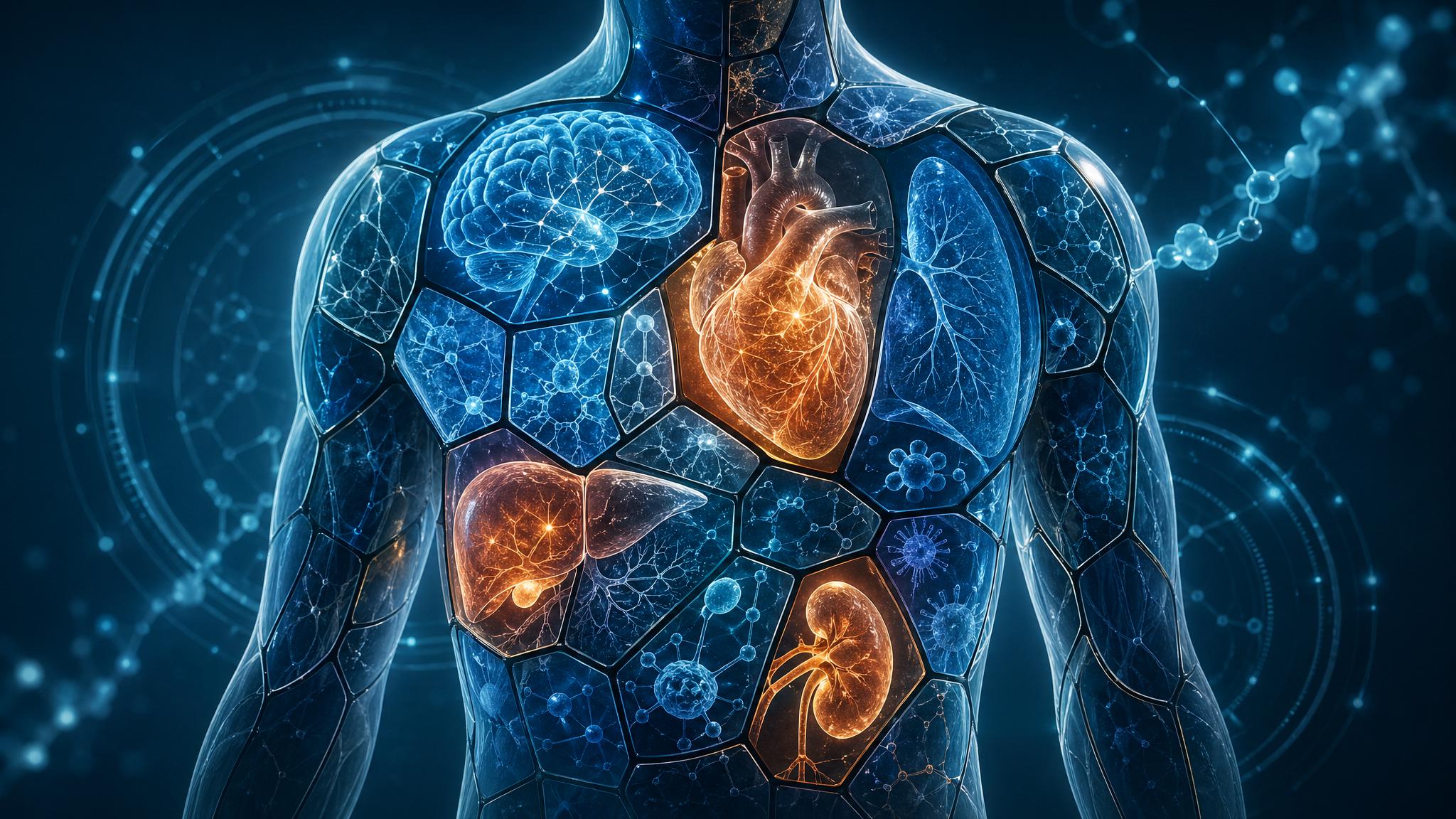
You will probably notice, even without trying, that they are not aging at one rate. The cardiovascular system might be holding up beautifully, the legs strong from a lifetime of walking, while memory has begun to soften. Or memory is sharp and the heart is weakening. Or the heart and brain are fine and the kidneys are years ahead of the rest of the body in their decline. We have all met the eighty-year-old whose body looks younger than her face, and the fifty-year-old whose joints already seem old.
For a long time, this everyday observation has lived just outside the formal science of aging. Researchers built theories around average decline, talked about lifespan and healthspan as singular numbers, and treated the body as a single decaying system slowly running down. But the evidence has been quietly piling up that this is the wrong picture.
In April 2026, the evolutionary biologists Pablo Burraco of the Doñana Biological Station in Spain and Jelle J. Boonekamp of the University of Glasgow published a preprint with a title that gets at the new picture directly: Mosaic Ageing: From Organ-Specific Decline to the Cause of Death. Their argument is at once simple and disorienting. The body does not fail all at once. It fails one organ at a time, almost always with a single critical system going first. The pace at which that fastest-aging organ breaks down, not the average pace of decline across the body, is what kills us.
If they are right, the goal of longevity medicine begins to shift in a useful and concrete way.
An old observation, finally formalized
The phrase “mosaic aging” was coined in 2010 by the Emory neurologist Lary Walker, in a paper that proposed treating the differential aging of organs as a fundamental feature of senescence rather than a quirk. Walker’s framing was elegant. Each person, he argued, experiences a unique constellation of changes that affect molecules, cells, organs, and systems, an idiosyncratic pattern of decline shaped by genetics, epigenetics, and the local environment of each tissue. He called this pattern the mosaic.
For most of the next decade, the idea sat in the literature as an interesting framing without a strong empirical foundation. It made intuitive sense; it was hard to prove rigorously. The tools to measure organ-specific aging at scale did not yet exist.
That has changed in the last three years, and the change has been dramatic.
In 2025, a Stanford-led team published a system of plasma proteomic aging clocks: blood-based biomarker panels that estimate the biological age of individual organs. By measuring a few thousand proteins circulating in the bloodstream, the team could assign each organ in the body, including the brain, heart, liver, kidneys, lungs, and immune system, its own biological age, distinct from the person’s chronological age. Some people had a brain ten years younger than their birth certificate suggested. Others had kidneys ten years older. Most had a striking spread, with at least one organ aging significantly faster than the rest.
In late 2025 and early 2026, follow-on work in Nature Aging validated this approach in a UK Biobank cohort of 43,616 individuals, with replication in Chinese and U.S. cohorts of nearly 5,000 more. The results were unambiguous. Organ-specific aging clocks predicted disease onset, progression, and mortality beyond what conventional clinical and genetic risk factors could capture. Among the eleven organs measured, accelerated brain aging carried the strongest link to mortality. Heart and kidney aging were close behind. The aging clocks were not telling the same story as a single number; they were telling eleven stories, and one of them mattered more than the others for any given person.
This is the empirical foundation Walker’s 2010 framing had been waiting for. Burraco and Boonekamp’s 2026 preprint is the theoretical scaffolding that ties it all together.
Why one organ matters more than the average
The core insight of the mosaic aging framework is borrowed, in spirit, from engineering. A bridge does not collapse because every component is at the average level of stress; it collapses when one critical component crosses its individual breaking point. The rest of the bridge is fine until it isn’t.
The body, Burraco and Boonekamp argue, works the same way. Death from natural causes in a person of advanced age is almost never the simultaneous failure of every system. It is, in the overwhelming majority of cases, the failure of one critical organ, with the rest of the body in varying states of compromise but not yet at threshold. A heart attack. A stroke. Pneumonia in lungs that have been getting weaker for years. Kidney failure. The body’s other systems, while not in great shape, were nowhere near failure.
Featured Partner
Invest in the Infrastructure Behind Modern Medicine
As healthcare expands beyond hospital walls, the buildings and campuses supporting that shift are generating compelling returns for investors who move early. The Healthcare Real Estate Fund offers qualified investors direct access to a curated portfolio of medical office, outpatient, and specialty care facilities.
Learn More →The implication is that the biological age of the body’s weakest organ, not the body’s average biological age, is the variable that most strongly predicts time of death. Two seventy-year-olds with identical “average” biological age can have radically different mortality risks if one of them has a brain ten years older than the rest of him and the other has uniformly intermediate aging across all organs. The first man’s clock is the brain. The second man’s clock is whichever organ happens to be slightly worse than the others, but no organ is dangerously ahead of its peers.
This reframes the goal of longevity medicine. It is no longer enough to reduce average wear and tear. The leverage is in identifying each individual’s fastest-aging organ and intervening there. A 0.3-year reduction in heart age, distributed across someone whose cardiovascular system is already in fine shape, may be worth less than a 0.3-year reduction concentrated in a kidney that is currently the patient’s weakest tile.
The mutational engine underneath
Why do organs age at different rates? The mosaic aging framework does not rest on any single answer, but a leading explanation, and the one that links this work to the broader Mosaic Body view we have been exploring, is the slow, organ-specific accumulation of somatic mutations.
We have written more broadly about the new science of somatic mosaicism elsewhere in this series. The basic point bears repeating here. Every cell in the body acquires DNA changes throughout life, in a pattern shaped by how often that tissue divides, what environmental insults it experiences, and how its repair machinery copes. Tissues with high turnover, like the lining of the gut or the blood-forming stem cells of the bone marrow, accumulate mutations differently from tissues with low turnover, like neurons. Tissues exposed to the outside world, like skin and the upper digestive tract, accumulate damage differently from internal tissues. Tissues in inflammatory environments mutate faster than tissues in quiet ones.
The result is that, by middle age, the human body is genuinely a patchwork of cellular populations evolving on different timelines, in different chemical environments, with different mutation burdens. A 2022 study from the Wellcome Sanger Institute that sequenced colonic crypts of sixteen mammalian species showed that the rate of somatic mutation per year scales inversely with lifespan with remarkable consistency: longer-lived species mutate slower. Whatever maintains low mutation rates in long-lived animals is doing real work to slow aging.
Inside any individual human body, the same logic applies between organs. The pancreas of a person with chronic high blood sugar accumulates damage at a different rate from the pancreas of someone with metabolic stability. The heart of a person living through chronic high-grade inflammation ages faster than the heart of a person who is not. The brain of a person under sustained sleep deprivation does its repair work less efficiently than the brain of a person who sleeps well. None of this is news in isolation. What is new is that we now have the tools to measure it at organ-level resolution, in living people, and to act on the results.
What the new measurement makes possible
The Burraco-Boonekamp framework is theoretical. The Stanford and UK Biobank organ aging clocks are empirical. The clinical translation of both is what is starting to arrive now.
Within the next five to ten years, we expect plasma proteomic aging panels to move from research-grade tests to clinically available ones, much as cholesterol panels and HbA1c moved from specialist tools to routine primary care over the second half of the twentieth century. Once organ-specific biological age is something a physician can order, three things become possible.
The first is risk stratification with a sharper edge than current tools allow. A patient whose cardiovascular age is twelve years ahead of their chronological age has a different short-term risk profile than a patient whose lung age is twelve years ahead, and a still different one from a patient whose brain age is the standout. Treatment decisions, including which preventive interventions to pursue, may change based on which organ is the weakest tile.
The second is the targeted use of interventions that, while broadly beneficial, are particularly suited to specific organs. Aerobic conditioning is most directly protective for the heart and brain. Resistance training is most directly protective for muscle, bone, and metabolic regulation. GLP-1 agonists appear to slow aging signatures in the cardiovascular and metabolic systems specifically. Senolytics, when they mature clinically, may have organ-specific patterns of benefit that we do not yet fully understand. Mosaic aging gives clinicians a vocabulary for matching the intervention to the tile.
The third, and most speculative, is the eventual emergence of organ-specific longevity therapies that aim to remove dangerous somatic mutations or eliminate clonally expanded mutated cells from a tissue before they tip the organ into failure. This is far off, but the vector of research is unmistakable. The Sanger Institute’s NanoSeq sequencing technology, the recent finding that somatic mutations in immune cells drive Hashimoto’s thyroiditis, and the broader Somatic Mosaicism Network’s reference catalogue all point in the same direction: organ-by-organ readouts, then organ-by-organ interventions.
What this means for your health
Mosaic aging is, despite its theoretical complexity, a profoundly empowering framework for ordinary decisions. Two ideas follow from it directly.
The first is that average matters less than spread. A person whose organs are all aging at roughly the same rate has a different prognosis from a person whose organs are aging at radically different rates, even if their average biological age is identical. Looking after every system, not just the one or two that conventional medicine measures most often (typically the heart and the metabolic system), is more important than the simple version of longevity advice has tended to suggest. The gut, the brain, the kidneys, the immune system, the liver, the lungs, all matter, and the one being neglected may be the one that ends up driving the timeline.
The second is that the foundational practices of healthspan are mosaic interventions. Whole-food nutrition reduces oxidative stress in the cardiovascular system, the liver, and the gut. Sleep restores DNA repair across every tissue. Resistance training preserves muscle and bone but also improves systemic insulin sensitivity, which protects pancreas, kidneys, and brain. Aerobic exercise builds cardiovascular reserve and increases the brain’s resistance to age-related decline. Stress regulation reduces the inflammatory tone in which somatic mutations preferentially accumulate. Strong social connection is one of the few interventions consistently associated with reduced all-cause mortality across virtually every organ system. The five foundational pillars of healthspan are not a single intervention. They are five interventions that act, in different proportions, across the entire mosaic.
If the Burraco-Boonekamp framework holds, the most consequential question a longevity-focused person can ask is not “how old is my body?” It is “which organ in my body is aging fastest, and what can I do about it?” Within a decade, that question will have a clinical answer. In the meantime, the foundational practices that broadly reduce somatic mutation, inflammation, and metabolic stress across all tissues are still the right place to focus, because they shore up the mosaic from beneath, before any single tile reaches its breaking point.
Aging, it turns out, is not a single number. It never was. It is a mosaic, and the goal is to keep every tile intact long enough that the picture, taken as a whole, holds together.
Frequently asked questions
What is mosaic aging?
Mosaic aging is the observation, formalized by neurologist Lary Walker in 2010 and extended by Burraco and Boonekamp in 2026, that organs and tissues in the body age at different rates rather than in lockstep. A person can have a young heart and an old kidney, or a young brain and an old liver, with each organ following its own biological clock shaped by genetics, environment, and the local accumulation of cellular damage.
Why does mosaic aging matter for lifespan?
Death in old age is almost never the simultaneous failure of every system. It is the failure of one critical organ, with the rest of the body in varying states of decline but not yet at the breaking point. This means the biological age of the fastest-aging organ, not the average biological age of the body, is the strongest predictor of mortality. Extending lifespan may depend more on protecting the weakest tile than on slowing aging across the body uniformly.
Can I measure how fast my individual organs are aging?
Research-grade plasma proteomic aging clocks now exist for at least eleven organs, validated in cohorts of nearly fifty thousand people in the UK, China, and the United States. These tests measure a few thousand proteins in the blood and assign each organ its own biological age. They are not yet routine clinical tools, but several are becoming available through specialized longevity clinics and direct-to-consumer testing platforms, and broader clinical availability is likely within five to ten years.
Which organs age fastest, and which matter most?
This varies dramatically between individuals. In population studies, accelerated brain aging is most strongly linked to all-cause mortality, with cardiovascular and kidney aging close behind. But for any given person, the answer depends on their unique genetic background, environmental exposures, lifestyle, and underlying disease risk. The point of organ-specific aging clocks is precisely to identify which organ is the personal weakest tile.
What causes organs to age at different rates?
The leading explanation involves the differential accumulation of somatic mutations, oxidative damage, and inflammatory exposure across tissues. Tissues with high cell turnover, high environmental exposure, or sustained inflammatory pressure accumulate cellular damage faster than protected tissues. Genetics also plays a role; some people inherit slower-aging variants of certain organs and faster-aging variants of others.
What can I do to protect my fastest-aging organ?
Until organ-specific aging tests become routinely available, the best strategy is the broad one: foundational practices that reduce somatic damage across all tissues. Whole-food nutrition, adequate sleep, regular movement combining aerobic and resistance training, stress regulation, and strong social connection each protect the entire mosaic. Once organ-specific testing is available, targeted interventions matched to the weakest tile will likely become the standard of personalized longevity medicine.
This piece is part of an ongoing series on the Mosaic Body, the emerging view of human biology in which acquired somatic mutations and tissue-specific cellular evolution shape health, disease, and aging. See also our pillar essay on why your cells are quietly mutating, and why it’s rewriting medicine, and our piece on somatic mutations and the new model of autoimmune disease.
References: Burraco, P. and Boonekamp, J. J. (2026). Mosaic Ageing: From Organ-Specific Decline to the Cause of Death. Authorea. DOI: 10.22541/au.177341917.74005102/v1. Walker, L. (2010). Mosaic aging. Medical Hypotheses. Wang, Q., et al. (2026). Organ-specific proteomic aging clocks predict disease and longevity across diverse populations. Nature Aging, 6:162-180.