Eko Duo: The Digital Stethoscope With Built In ECG for Dual Cardiac Screening
The most advanced digital stethoscope available, combining AI powered heart sound analysis with a single lead ECG to screen for murmurs, AFib, and structural heart disease simultaneously during routine examination.
A cardiac examination has historically been a two step process: auscultation (listening to heart sounds with a stethoscope) and electrocardiography (recording the heart’s electrical activity with an ECG). These two modalities capture fundamentally different dimensions of cardiac function. The stethoscope detects mechanical abnormalities: the turbulent blood flow that produces murmurs, the abnormal filling sounds that indicate heart failure, the clicks and rubs that suggest valvular or pericardial disease. The ECG detects electrical abnormalities: rhythm disturbances, conduction delays, ischemic changes, and chamber enlargement patterns.
Historically, these assessments required separate instruments, separate workflows, and often separate clinical encounters. A clinician might hear something concerning during auscultation and order an ECG, or an ECG might suggest structural disease that prompts echocardiography. The Eko Duo collapses this two step process into a single instrument that captures both heart sounds and electrical activity simultaneously, with AI algorithms analyzing both data streams in real time.
This is not incremental improvement. It represents a fundamental expansion of what a stethoscope examination can accomplish.
What Is the Eko Duo?
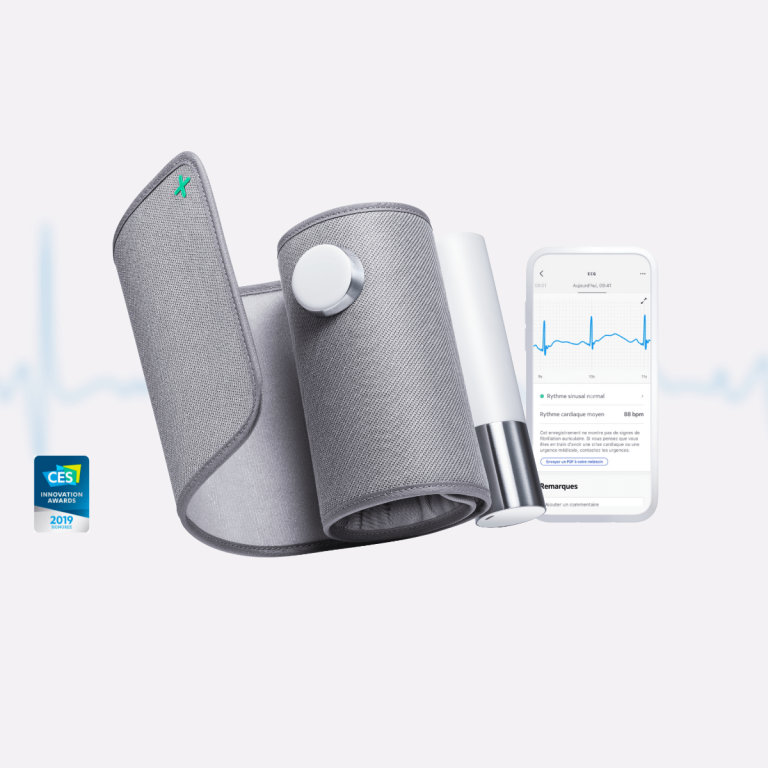
The Eko Duo is an FDA cleared digital stethoscope with an integrated single lead ECG that simultaneously records heart sounds and an electrocardiogram during cardiac examination. The device features a stethoscope chest piece with embedded ECG electrodes; when placed on the patient’s chest, it captures a phonocardiogram (digital heart sound recording) and a single lead ECG tracing in a single session.
The device provides 40x sound amplification with active noise cancellation for heart sound clarity, plus AI powered analysis for three screening functions: cardiac murmur detection, atrial fibrillation detection via heart sound rhythm analysis, and ECG based rhythm analysis. All three AI algorithms hold FDA 510(k) clearance for their respective screening functions.
Recordings sync via Bluetooth to the Eko app, which displays the simultaneous phonocardiogram and ECG tracing, presents AI screening results, and enables sharing with specialists for remote review. An optional Eko Premium subscription ($9.99 per month) adds enhanced storage and reporting. Core AI functions work without subscription. The Duo retails at $399 and is confirmed HSA and FSA eligible.
The Science Behind Simultaneous Auscultation and ECG
The clinical value of simultaneous heart sound and ECG recording lies in the complementary information each modality provides. Timing cardiac events against the ECG tracing allows precise characterization of murmurs: a systolic murmur (occurring between S1 and S2, coinciding with ventricular contraction on ECG) suggests different pathology than a diastolic murmur (occurring between S2 and the next S1, during ventricular filling). The ECG provides the timing reference that transforms auscultation from a qualitative assessment into a semi quantitative one.
This approach has been used in clinical cardiology for decades through formal phonocardiography, but historically required dedicated laboratory equipment that was impractical for routine clinical use. The Eko Duo brings this capability to a portable device that can be used during any clinical encounter.
The dual modality approach also provides redundant screening for atrial fibrillation. The heart sound algorithm detects the irregularly irregular rhythm pattern acoustically, while the ECG algorithm detects the same rhythm disturbance electrically. When both modalities agree, clinical confidence in the screening result increases. When they disagree, the discrepancy itself provides useful clinical information that warrants further evaluation.
The BASEL Wearable Study (Mannhart et al., 2023, JACC: Clinical Electrophysiology) validated consumer ECG devices for AFib detection and found that no single modality achieves perfect sensitivity. Combining acoustic and electrical analysis represents a logical approach to improving screening yield, though published validation data for the specific combination implemented in the Eko Duo is still accumulating.
What the Eko Duo Does Well
The Duo’s primary strength is integration. Capturing heart sounds and ECG simultaneously, from the same chest placement, during the same examination moment, eliminates the workflow separation that traditionally exists between auscultation and electrocardiography. For busy clinicians, this integration means more comprehensive cardiac screening without additional time investment.
The simultaneous display of phonocardiogram and ECG tracing in the Eko app provides a visualization that enhances clinical understanding. Seeing the relationship between electrical events (P waves, QRS complexes) and mechanical events (heart sounds, murmurs) in real time is an educational and diagnostic tool that improves pattern recognition over time.
All three AI screening functions are FDA cleared, providing a regulatory validation layer that distinguishes the Duo from uncleaned digital stethoscopes. The murmur detection AI compensates for the auscultation skill variability that affects non specialist clinicians, while the dual modality AFib screening (acoustic and electrical) provides redundant detection pathways.
Featured Partner
Invest in the Infrastructure Behind Modern Medicine
As healthcare expands beyond hospital walls, the buildings and campuses supporting that shift are generating compelling returns for investors who move early. The Healthcare Real Estate Fund offers qualified investors direct access to a curated portfolio of medical office, outpatient, and specialty care facilities.
Learn More →For telehealth applications, the Duo is particularly powerful. A clinician can perform a remote cardiac examination by sharing both the heart sound recording and the ECG tracing with a consulting cardiologist, providing the specialist with the same two data streams they would obtain from an in person examination. This capability is transformative for rural and underserved settings where specialist access is limited.
Pricing, Access, and Practical Realities
The Eko Duo retails at $399, a $100 premium over the Eko Core 500 ($299). The premium buys the integrated ECG capability. For clinicians who would otherwise use a stethoscope and a separate ECG device, the consolidated approach may represent a net reduction in equipment cost and workflow complexity.
No subscription is required for core AI screening functions (murmur detection, AFib detection via both acoustic and ECG analysis). The optional Eko Premium subscription at $9.99 per month adds unlimited cloud storage, advanced reporting, and additional analytical features.
The Duo holds FDA 510(k) clearance for all three AI screening functions: murmur detection, acoustic AFib detection, and ECG based rhythm analysis. This comprehensive clearance validates the device as a clinically legitimate screening platform. The device is confirmed HSA and FSA eligible.
Battery life supports approximately 9 hours of continuous clinical use, and the device charges via USB C. The stethoscope functions as an amplified digital stethoscope even without Bluetooth app connection, but AI screening and ECG recording require the app.
Who the Eko Duo Is Best For
The Duo is the optimal choice for clinicians who want the most comprehensive cardiac screening tool available in a single handheld device. Cardiologists conducting routine follow up examinations can capture both acoustic and electrical data in one placement. Primary care physicians performing annual physicals gain murmur and AFib screening capabilities that augment their clinical assessment.
Internal medicine physicians, hospitalists, and emergency medicine physicians who encounter cardiac patients across diverse clinical settings will find the portability and dual modality valuable. Nurse practitioners and physician assistants who may have less extensive auscultation training benefit disproportionately from the AI augmentation that compensates for experience gaps.
Telehealth providers who need to perform remote cardiac assessment should strongly consider the Duo. The ability to share both heart sound recordings and ECG tracings with remote specialists creates a clinical consultation pathway that closely replicates in person cardiac examination.
Clinicians who need only stethoscope AI without ECG should save $100 by choosing the Core 500. Consumers seeking personal AFib monitoring should choose a dedicated consumer ECG device (AliveCor KardiaMobile, Apple Watch) rather than a clinical stethoscope. The Duo is designed for clinical cardiac examination, not consumer self monitoring.
How the Eko Duo Compares
Against the Eko Core 500 ($299), the Duo adds single lead ECG capability at a $100 premium. Both share the same AI stethoscope platform with murmur and acoustic AFib detection. The Duo’s ECG provides an additional screening modality and a shareable electrical tracing. For clinicians who value the ECG data stream, the premium is justified; for those focused on auscultation augmentation alone, the Core 500 suffices.
Against the Withings BPM Core ($249.95), which also combines stethoscope and ECG capabilities, the Duo provides a clinical grade instrument designed for provider use with significantly better acoustic quality, 40x amplification, AI screening, and a form factor suited to clinical examination. The BPM Core is a consumer blood pressure monitor with embedded stethoscope and ECG; the Duo is a professional clinical instrument. These serve fundamentally different use cases.
Against traditional stethoscope plus portable ECG combinations (e.g., Littmann Classic III at $100 plus AliveCor KardiaMobile at $99), the Duo consolidates both functions at $399 with the addition of AI screening. The combined traditional approach costs roughly $200 but requires two separate workflows and lacks AI augmentation. The Duo provides integration and AI at approximately double the combined cost.
Limitations and Open Questions
The Duo is a clinical instrument that requires proper stethoscope technique for meaningful results. Incorrect placement, excessive pressure, or movement during recording will degrade both the heart sound and ECG quality. The device does not compensate for fundamental examination skill deficiencies beyond what the AI augmentation provides.
The single lead ECG provides limited diagnostic information compared to a standard 12 lead clinical ECG. While sufficient for rhythm screening and AFib detection, it cannot reliably identify ischemic changes, bundle branch blocks, or many structural abnormalities that require multi lead assessment. The ECG function supplements, but does not replace, standard electrocardiography.
The AI screening algorithms flag potential abnormalities but do not provide specific diagnoses. A “murmur detected” result requires echocardiographic follow up; an “AFib detected” result requires clinical ECG confirmation. The device increases screening sensitivity but does not reduce the need for confirmatory diagnostic testing.
At $399, the Duo represents a significant investment for an individual clinician, though it is competitive with premium traditional stethoscopes and substantially less expensive than dedicated clinical ECG equipment. The optional subscription adds ongoing cost for practices that want full feature access.
What This Means for Your Health
The Eko Duo represents a meaningful advance in how cardiovascular disease, one of the Four Villains, is detected during routine clinical encounters. By combining AI augmented heart sound analysis with ECG recording in a single instrument, it increases the probability that murmurs, atrial fibrillation, and other cardiac abnormalities are identified during the clinical encounters that are already happening, rather than requiring separate screening appointments.
Within Healthcare Discovery‘s editorial framework, the Duo exemplifies the convergence of artificial intelligence and clinical medicine that is central to the platform’s thesis. The stethoscope has been listening to hearts for over 200 years. The Eko Duo is the first version that listens and analyzes simultaneously, providing AI powered cardiac screening during the most fundamental clinical interaction: the physical examination.
For patients, the practical implication is straightforward: ask your clinician whether they use an AI augmented stethoscope, and if so, request that your cardiac examination include both acoustic and ECG screening. Early detection of murmurs and rhythm disturbances during routine visits is one of the most effective pathways to catching cardiovascular disease before it progresses to symptomatic, harder to treat stages.
Explore the full wearable guide
See how the Eko Duo compares with smart rings, watches, ECG devices, and other connected health hardware across the full Healthcare Discovery wearables guide.
Frequently Asked Questions
Is the Eko Duo FDA cleared?
Yes. The Eko Duo holds FDA 510(k) clearance for three AI screening functions: cardiac murmur detection via heart sound analysis, atrial fibrillation detection via acoustic rhythm analysis, and ECG based rhythm analysis. This makes it one of the most comprehensively cleared digital cardiac screening instruments available.
What is the difference between the Eko Core 500 and the Eko Duo?
The Core 500 ($299) is a digital stethoscope with AI murmur and AFib detection via heart sound analysis. The Duo ($399) adds a single lead ECG that records simultaneously with the heart sound, providing both acoustic and electrical cardiac data in a single device. The $100 premium buys the integrated ECG capability and dual modality AFib screening.
Does the Eko Duo require a subscription?
Core AI screening functions (murmur detection, AFib detection, ECG analysis) work without a subscription. The optional Eko Premium subscription at $9.99 per month adds unlimited cloud storage, advanced reporting features, and additional analytical capabilities. Most clinicians can use the essential screening functions without subscribing.
Can consumers use the Eko Duo at home?
The Eko Duo is available for consumer purchase and can technically be used at home. However, it is designed for clinical cardiac examination and requires proper stethoscope placement technique and basic cardiac anatomy knowledge for meaningful results. Most consumers seeking home cardiac screening would be better served by a dedicated consumer ECG device (AliveCor KardiaMobile) or a blood pressure monitor with ECG (Omron Complete).
Is the Eko Duo HSA or FSA eligible?
Yes. The Eko Duo is confirmed HSA and FSA eligible with a Letter of Medical Necessity. At $399, it can be purchased with pre tax healthcare dollars, reducing the effective cost for eligible clinicians and consumers.