AliveCor KardiaMobile 6L: The Most Clinically Advanced Consumer ECG Device
A pocket sized, six lead ECG that captures half the diagnostic information of a full clinical 12 lead, FDA cleared for six arrhythmia determinations, and priced at $149 with no subscription required.
A standard clinical 12 lead ECG views the heart’s electrical activity from 12 different angles, creating a comprehensive map that cardiologists use to diagnose everything from atrial fibrillation to myocardial infarction. Most consumer ECG devices capture a single lead (Lead I), which is sufficient for detecting the most common rhythm disturbances but provides only one viewing angle of a complex three dimensional electrical event. The gap between a single lead and a full 12 lead recording represents a significant limitation in how much diagnostic information a consumer device can provide.
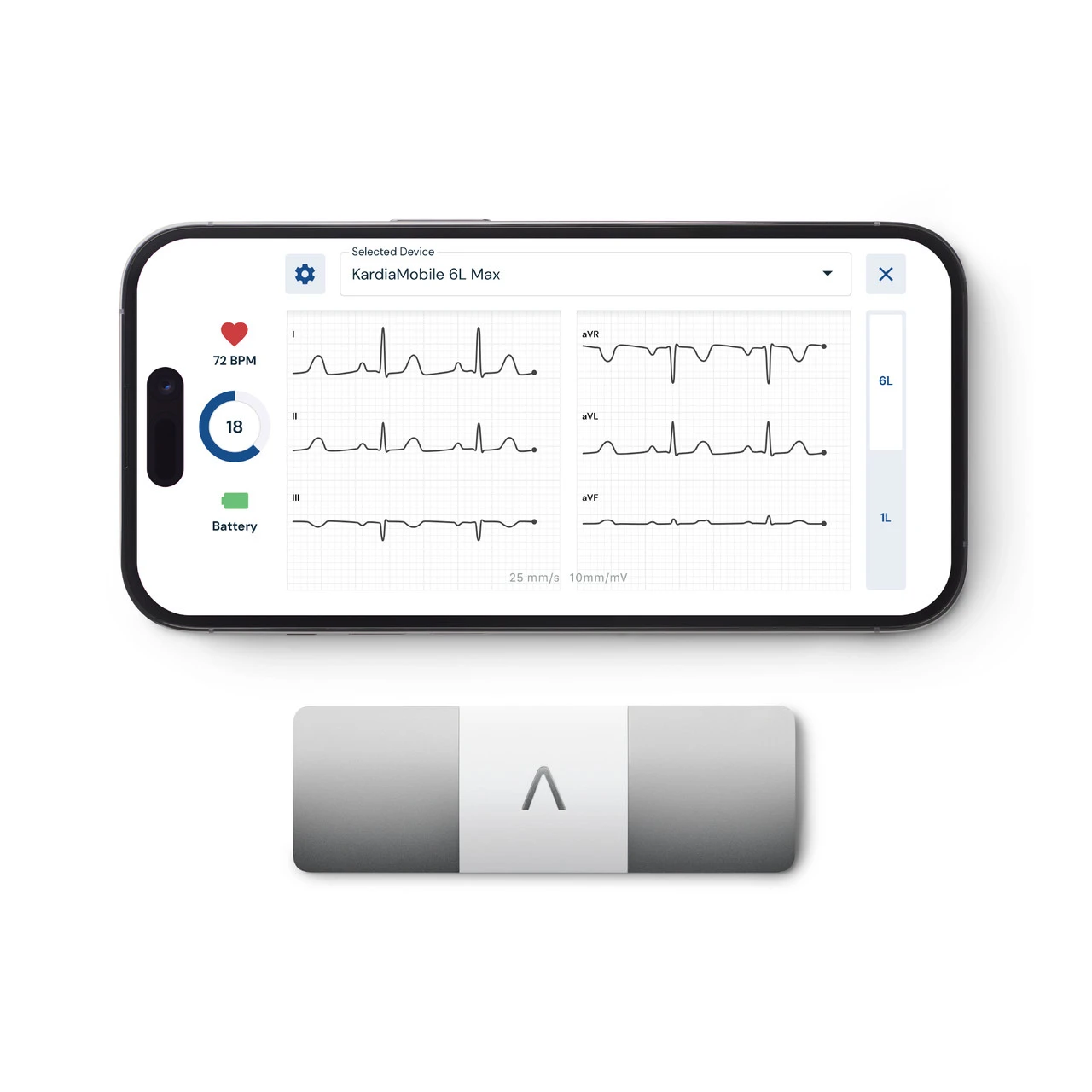
The AliveCor KardiaMobile 6L narrows that gap by capturing six of the twelve standard leads in a single 30 second recording. Leads I, II, III, aVR, aVL, and aVF, collectively known as the limb leads, provide multiple viewing angles of the heart’s frontal plane electrical activity. This additional perspective enables detection of conditions that single lead recordings may miss, including certain patterns of premature ventricular contractions, conduction abnormalities, and more nuanced rhythm characteristics.
In the BASEL Wearable Study published in 2023 in JACC: Clinical Electrophysiology, the standard AliveCor KardiaMobile achieved 79% sensitivity and 69% specificity for atrial fibrillation detection. The 6L model provides the same detection algorithms for AFib plus the additional leads that give physicians substantially more diagnostic data to work with when reviewing the recordings.
What Is the AliveCor KardiaMobile 6L?
The AliveCor KardiaMobile 6L is a portable, smartphone connected ECG device that records a six lead electrocardiogram in 30 seconds. The device is slightly larger than the standard KardiaMobile but still fits comfortably in a pocket, roughly the size of a business card holder. It features two finger electrodes on the top surface and a leg electrode on the bottom, which contacts the user’s thigh or knee during recording to create the multi lead configuration.
To record an ECG, the user opens the Kardia app, places two fingers from each hand on the upper electrodes while resting the bottom electrode against their left leg, and waits 30 seconds. The app immediately analyzes the six lead recording and provides rhythm classification. The FDA cleared determinations include atrial fibrillation, bradycardia, tachycardia, normal sinus rhythm, premature ventricular contractions (PVCs), sinus rhythm with supraventricular ectopy (SVE), and unclassified or unreadable recordings.
The 6L holds FDA 510(k) clearance and is the most clinically advanced consumer ECG device currently available. It syncs via Bluetooth to the Kardia app on iOS and Android, stores recordings with timestamps, and enables one tap PDF sharing with healthcare providers. The optional KardiaCare subscription ($9.99 per month or $99 per year) adds enhanced reporting and unlimited storage. Core ECG functionality requires no subscription. The device retails at $149 and is confirmed HSA and FSA eligible.
The Science Behind Multi Lead ECG Screening
The value of a multi lead ECG over a single lead recording lies in the additional diagnostic information each lead provides. A single lead (Lead I) views the heart from a single electrical angle, analogous to photographing a building from one side. Six leads provide six angles, enough to assess the heart’s electrical axis, identify certain conduction delays, and distinguish between similar appearing rhythms that a single lead might classify identically.
For atrial fibrillation detection specifically, six leads do not dramatically change sensitivity compared to single lead recording, because AFib’s hallmark features (irregular rhythm, absent P waves) are identifiable from any standard lead. Where six leads add clinical value is in characterizing the overall cardiac electrical landscape: identifying PVCs (which a single lead can detect but cannot always distinguish from supraventricular beats), assessing axis deviation (which may suggest underlying structural disease), and providing multiple perspectives on P wave morphology and QRS duration.
A 2020 machine learning study by Lown et al. in PLoS One demonstrated that even single channel rhythm data can achieve 100% sensitivity and 97.6% specificity for AFib detection when processed with sophisticated algorithms. The clinical advantage of six leads is not necessarily better AFib detection; it is the additional conditions the device can screen for and the richer data it provides to the reviewing physician.
For cardiologists reviewing consumer ECG recordings, six lead tracings are substantially more useful than single lead tracings. They approach the information density of the first half of a standard 12 lead ECG and enable clinical assessments that single lead recordings simply cannot support. This is why many cardiologists who accept consumer ECG data prefer the 6L recordings over single lead alternatives.
What the AliveCor KardiaMobile 6L Does Well
The 6L’s primary differentiator is the clinical depth of its recordings. No other consumer device captures a six lead ECG in a portable form factor. The additional leads transform the device from a rhythm screening tool into something approaching a basic diagnostic instrument, though it remains a screening device by regulatory classification.
FDA clearance for PVC detection is a meaningful addition over single lead devices. Premature ventricular contractions are the most common ventricular arrhythmia, and while isolated PVCs are usually benign, frequent PVCs (greater than 10% of total heartbeats) can indicate underlying cardiac pathology and may warrant further evaluation. The ability to detect and document PVCs outside a clinical setting adds practical value for patients with palpitations or known ventricular ectopy.
The recording process, while slightly more involved than the standard KardiaMobile (requiring leg contact), still takes only 30 seconds and requires no external equipment beyond a compatible smartphone. The device uses Bluetooth connectivity, which eliminates the ambient noise sensitivity of the standard model’s ultrasonic communication. Battery life using a coin cell battery extends approximately 12 months.
Featured Partner
Invest in the Infrastructure Behind Modern Medicine
As healthcare expands beyond hospital walls, the buildings and campuses supporting that shift are generating compelling returns for investors who move early. The Healthcare Real Estate Fund offers qualified investors direct access to a curated portfolio of medical office, outpatient, and specialty care facilities.
Learn More →AliveCor’s reputation among cardiologists is strongest for the 6L. Many electrophysiologists and general cardiologists specifically recommend the 6L to patients because its six lead recordings provide enough data for meaningful clinical interpretation, approaching the utility of an ambulatory event monitor without the prescription, rental fees, or limited wear duration of traditional clinical monitors.
Pricing, Access, and Practical Realities
The AliveCor KardiaMobile 6L retails at $149 with no subscription requirement for core ECG recording and arrhythmia detection. The optional KardiaCare subscription adds enhanced features at $9.99 per month or $99 per year. First year total cost ranges from $149 (device only) to $249 (device plus annual subscription).
At $149, the 6L is $50 more than the standard KardiaMobile and significantly less expensive than any smartwatch with ECG capability. For the specific use case of on demand, multi lead ECG recording with maximum clinical utility, it is the best value available. The incremental $50 over the standard model purchases substantially more diagnostic information per recording.
The device is confirmed HSA and FSA eligible with a Letter of Medical Necessity. FDA 510(k) clearance covers six arrhythmia determinations including PVCs and SVE, which the standard KardiaMobile does not detect. This broader detection capability and the richer six lead data justify the premium for patients with clinical ECG monitoring needs.
The 6L uses a coin cell battery (CR2032) rated for approximately 12 months. Replacement batteries cost under $5. The device is compatible with most recent iOS and Android smartphones via Bluetooth.
Who the AliveCor KardiaMobile 6L Is Best For
The 6L is best suited for patients with diagnosed cardiac conditions who need ongoing rhythm monitoring with maximum diagnostic depth. Patients with paroxysmal atrial fibrillation, known PVCs, or unexplained palpitations will benefit from the six lead capability. Patients whose cardiologists have recommended event monitoring between formal Holter monitor prescriptions represent a core audience.
Clinicians who prescribe consumer ECG devices to patients should consider the 6L as the default recommendation when budget allows. The additional leads meaningfully increase the clinical utility of each recording, making physician review more productive and reducing the ambiguity that accompanies single lead tracings.
Health conscious adults who want the most thorough personal ECG available, even without a specific cardiac diagnosis, will find the 6L provides peace of mind backed by genuine clinical capability. Biohackers and quantified self enthusiasts who track HRV and resting heart rate can use the 6L’s detailed ECG data to complement their broader cardiovascular monitoring stack.
Users who want continuous, passive rhythm monitoring should pair the 6L with a smartwatch ECG device. The 6L requires intentional recording sessions and cannot detect events between recordings. Users on a strict budget who need only AFib screening may find the standard KardiaMobile ($99) adequate for their needs.
How the AliveCor KardiaMobile 6L Compares
Against the standard AliveCor KardiaMobile ($99), the 6L provides five additional ECG leads and PVC/SVE detection at a $50 premium. The recording process is slightly more involved (requiring leg contact), but the clinical information gained is substantial. For patients with simple AFib screening needs, the standard model suffices. For patients who want the most useful recordings for physician review, the 6L is worth the upgrade.
Against the Apple Watch Series 9 ECG, the 6L provides six leads versus one, PVC detection versus AFib only, and generates recordings that cardiologists generally find more clinically useful. The Apple Watch provides continuous passive monitoring that the 6L cannot replicate. These devices serve complementary functions: the Apple Watch for 24/7 rhythm surveillance, the 6L for detailed on demand recordings when symptoms occur or when maximum diagnostic data is needed.
Against the Withings Body Scan, which also captures a 6 lead ECG, the KardiaMobile 6L offers greater portability and a more established clinical validation track record for its ECG algorithms. The Body Scan embeds its 6 lead ECG into a smart scale, adding body composition and other measurements but limiting portability. The 6L can be used anywhere, anytime; the Body Scan requires standing on the scale at home.
Limitations and Open Questions
Like all on demand ECG devices, the 6L cannot detect events that occur between recording sessions. Paroxysmal arrhythmias, by definition intermittent, may be missed if episodes do not coincide with active recordings. Users must be disciplined about recording during symptoms to maximize the device’s diagnostic yield.
The recording process requires three point contact (two fingers plus leg), which is slightly more complex than the single lead model’s two finger setup. Some users, particularly elderly patients with mobility limitations, may find the leg contact position awkward. The device can alternatively be placed on the left ankle instead of the thigh, which may be easier for some users.
While six leads provide substantially more information than one lead, they still represent only half of a standard 12 lead ECG. The precordial (chest) leads (V1 through V6) are absent, which means the 6L cannot assess the heart’s horizontal plane electrical activity. Conditions like ST elevation myocardial infarction, which are most reliably identified in precordial leads, cannot be screened for with the 6L.
The device has no display and requires a smartphone for all functionality. Users without compatible smartphones cannot use the device. In contrast, some competing devices (like the Omron Complete) include built in displays for immediate feedback without phone dependency.
What This Means for Your Health
Cardiovascular disease is one of the Four Villains, and cardiac arrhythmias are among its most treacherous manifestations: silent, unpredictable, and carrying consequences disproportionate to their subtlety. A single undetected AFib episode can cause a stroke. A pattern of frequent PVCs can indicate underlying structural disease. The AliveCor KardiaMobile 6L puts the most clinically advanced consumer ECG technology in your pocket, providing six leads of diagnostic data that rival the information density of a clinical event monitor.
Within Healthcare Discovery‘s Five Pillars framework, the 6L supports the Movement pillar (capturing cardiac response to exercise and detecting exercise induced arrhythmias), the Breathwork pillar (documenting how stress and breathing patterns affect heart rhythm), and the Sleep pillar (recording symptoms that occur during or upon waking from sleep). It serves the Mindset pillar by transforming cardiac uncertainty into documented data, replacing “I think something happened” with “here is the six lead recording from when it happened.”
If your cardiologist has recommended personal ECG monitoring, the 6L should be your first consideration. If you have risk factors for cardiac arrhythmias and want the most thorough consumer screening tool available, the 6L provides clinical depth that justifies its modest $149 price. Use it when symptoms occur, use it at regular intervals for baseline comparison, and share every recording with your healthcare team. The data you capture between clinical visits may be the most valuable diagnostic information your cardiologist receives.
Explore the full wearable guide
See how AliveCor KardiaMobile 6L compares with smart rings, watches, ECG devices, and other connected health hardware across the full Healthcare Discovery wearables guide.
Frequently Asked Questions
What is the difference between the KardiaMobile and KardiaMobile 6L?
The standard KardiaMobile captures a single lead ECG (Lead I) using two finger electrodes for $99. The KardiaMobile 6L captures six leads (I, II, III, aVR, aVL, aVF) using two finger electrodes plus a leg contact point for $149. The 6L additionally detects premature ventricular contractions (PVCs) and supraventricular ectopy (SVE), which the single lead model cannot identify. Both are FDA cleared and require no subscription for core functionality.
Is the AliveCor KardiaMobile 6L FDA cleared?
Yes. The KardiaMobile 6L holds FDA 510(k) clearance for six arrhythmia determinations: atrial fibrillation, bradycardia, tachycardia, normal sinus rhythm, PVCs, and sinus rhythm with SVE. It is the most clinically advanced consumer ECG device currently available and produces recordings that approach the diagnostic utility of the limb lead portion of a standard 12 lead clinical ECG.
Can the KardiaMobile 6L detect a heart attack?
No. The 6L captures the six limb leads of a standard ECG but does not capture the six precordial (chest) leads. ST elevation myocardial infarction (heart attack) is most reliably identified in the precordial leads. While some limb lead changes may suggest ischemia, the 6L is not designed or cleared for heart attack detection. Any chest pain or suspected cardiac emergency requires immediate emergency medical services, not a consumer ECG device.
Does the KardiaMobile 6L require a subscription?
No. Core ECG recording and arrhythmia detection work without any subscription. The optional KardiaCare subscription ($9.99/month or $99/year) adds medication tracking, monthly heart health reports, unlimited ECG storage, and cardiologist review of flagged recordings. Most users can use the device effectively without subscribing.
How do I share KardiaMobile 6L recordings with my doctor?
The Kardia app generates a PDF report for each recording that includes the full six lead ECG tracing, heart rate, duration, and algorithmic rhythm classification. You can share this PDF via email, text message, or print directly from the app. Many cardiologists are familiar with AliveCor recordings and accept them as clinically useful screening data. Some physicians may have a direct portal integration available through the KardiaPro platform.