FDA AI in Cardiology: Heart Monitoring Moves From the Clinic to the Body
The heart has always been one of medicine’s most measurable organs. It produces electricity. It makes sound. It changes rhythm. It pushes blood through vessels that can be imaged, modeled, and monitored. Long before artificial intelligence entered the conversation, cardiology had already built a culture around signals.
That makes heart care a natural second front for regulated medical AI.
In the current FDA artificial-intelligence-enabled medical device CSV, cardiovascular devices account for 136 entries. That places cardiology far behind radiology, which dominates the list, but well ahead of most other specialties. The reason is not mysterious. Cardiology sits at the intersection of structured data, urgent clinical risk, chronic disease, remote monitoring, and consumer health technology.
It is also where the boundary between hospital medicine and everyday life is beginning to blur.
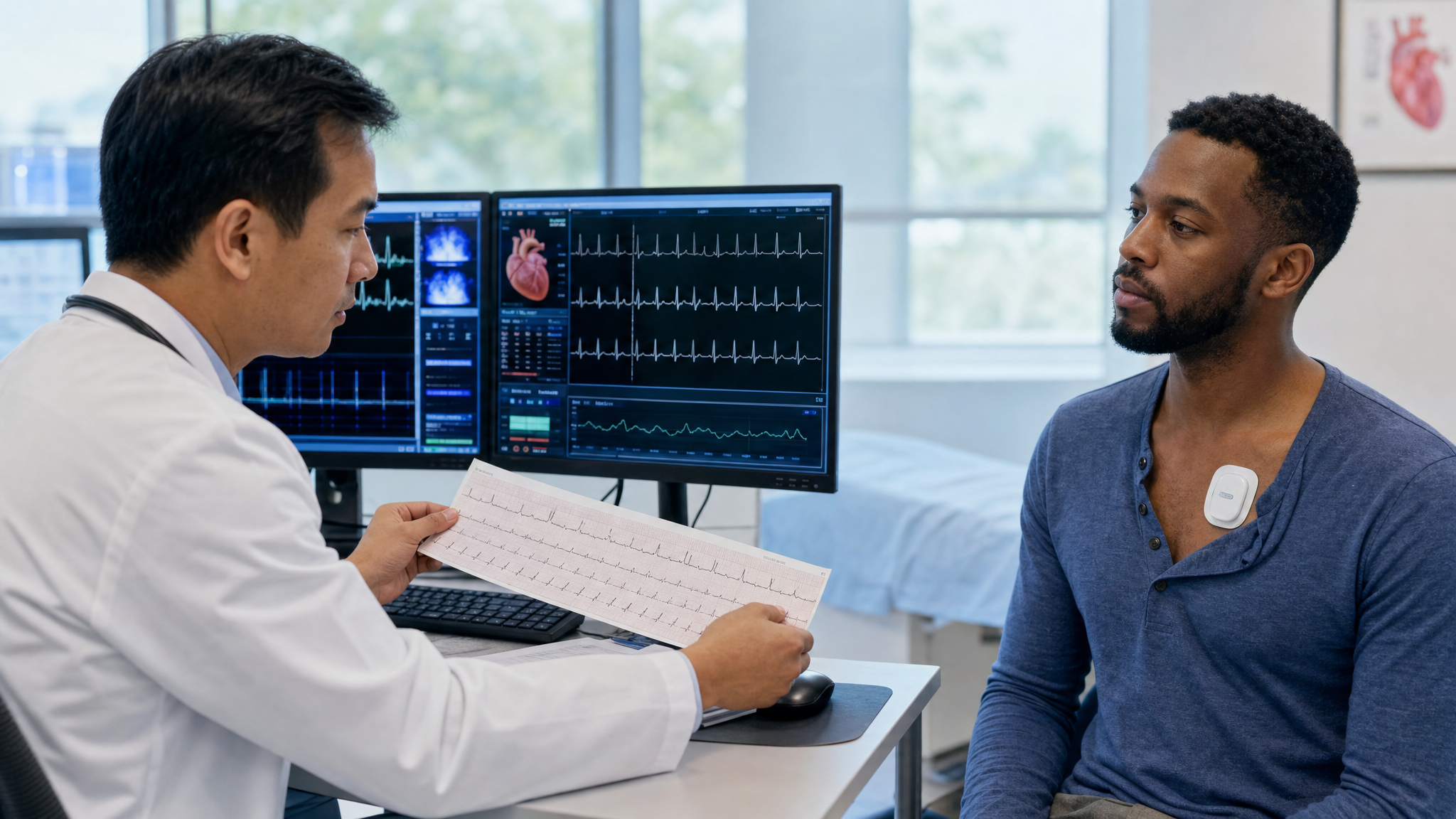
A person can now wear a watch that records an ECG, a patch that tracks rhythm over days, a ring or wrist device that detects physiologic patterns, or a connected sensor that sends signals back into a clinical system. Some of those tools live mostly in the wellness world. Others have FDA-authorized functions. Some appear on the FDA’s AI-enabled medical device list because artificial intelligence or machine learning is part of a regulated medical use.
This is the cardiology chapter of the broader FDA AI medical devices story: not one giant diagnostic machine, but many smaller intelligence layers watching the heart from different angles.
Other parts of that same cluster now trace AI through neurology, gastroenterology-urology and endoscopy, and anesthesiology’s sleep and respiratory category, all anchored to the main pillar page.
The Heart Became a Data Stream
Cardiology is built around patterns. A twelve-lead ECG can show electrical clues about rhythm, conduction, ischemia, prior injury, and chamber strain. A Holter monitor can catch intermittent arrhythmias that vanish during a clinic visit. An echocardiogram can reveal valve disease, ejection fraction, wall motion, chamber size, and hemodynamics. A CT-derived coronary model can help evaluate blood flow. A heart sound can carry information about murmurs, valves, and flow.
AI fits into cardiology because these signals are both meaningful and messy. The heart produces enormous amounts of information, but much of it arrives as waves, intervals, images, sounds, and time-series patterns. Those are exactly the kinds of data modern algorithms can help sort, classify, and prioritize.
The current FDA list reflects that range. Recent cardiovascular entries include eMurmur Heart AI from CSD Labs, Assert-IQ from Abbott, DeepRhythmAI from Medicalgorithmics, CardioVision from iCardio.ai, Bunkerhill ECG-EF from BunkerHill Health, Volta AF-Xplorer II from Volta Medical, Apple’s Hypertension Notification Feature, Eko Foundation Analysis Software with Transformers, the Cardiologs Holter Platform from Philips France Commercial, Anumana’s ECG-AI Low Ejection Fraction algorithm, HeartFlow Analysis, Tempus ECG-Low EF, Withings ECG App, Empatica Health Monitoring Platform, Fitbit’s Loss of Pulse Detection, and AliveCor’s Impala.
That list spans several worlds at once: clinical ECG interpretation, remote rhythm monitoring, heart-sound analysis, echocardiography, coronary modeling, wearable notifications, and patient monitoring platforms. Cardiology AI is not one product category. It is a convergence of signals.
From the Clinic to the Body
Radiology AI usually lives inside imaging rooms, scanners, workstations, and specialist workflows. Cardiology AI often moves closer to the body.
That difference matters. A CT scan happens at a particular moment in a particular facility. A heart rhythm can misbehave at 2:00 a.m., during a run, after a poor night of sleep, while sitting at a desk, or in the middle of an ordinary day. Atrial fibrillation can be intermittent. Blood pressure can drift. heart failure can worsen gradually. A dangerous rhythm can appear briefly and disappear before anyone sees it.
This is why cardiac monitoring has pushed so aggressively beyond the clinic. Watches, patches, portable ECG devices, implantable monitors, connected stethoscopes, and remote patient monitoring platforms all attempt to solve the same problem: the heart does not perform on command.
AI enters because continuous or repeated measurement produces too much raw signal for simple human review. A patch may record days of ECG data. A wearable may collect months of physiologic trends. A remote system may monitor many patients at once. The useful signal may be rare, subtle, or buried in noise.
Featured Partner
Invest in the Infrastructure Behind Modern Medicine
As healthcare expands beyond hospital walls, the buildings and campuses supporting that shift are generating compelling returns for investors who move early. The Healthcare Real Estate Fund offers qualified investors direct access to a curated portfolio of medical office, outpatient, and specialty care facilities.
Learn More →The promise is not that software replaces the cardiologist. It is that software can help find the moment worth a cardiologist’s attention.
The Consumer Overlap Is Real
Cardiology is also the medical AI category most visible to ordinary people.
Apple’s irregular rhythm notification and atrial fibrillation history features, Withings ECG tools, Fitbit’s loss-of-pulse detection, AliveCor’s Kardia-related ecosystem, Empatica’s monitoring platform, and other connected devices have helped normalize the idea that cardiac signals can be captured outside the clinic. Not every feature is AI-enabled. Not every wearable function is a medical device. But the overlap is impossible to ignore.
The shape of the device can be misleading. A watch may look like a consumer product while one specific feature carries FDA authorization. A medical patch may look simple while producing clinically important rhythm data. A stethoscope may feel old-fashioned while its software listens with algorithmic help. A blood-pressure or hypertension notification may sit inside a broader consumer platform but still be tied to a regulated function.
The important distinction is not whether the product is familiar. It is what the specific function is authorized to do.
That is where public understanding often breaks down. A health tracker can be useful without being diagnostic. A notification can identify a pattern without proving a disease. An ECG feature can capture a signal without replacing a full clinical evaluation. A monitoring platform can assist care without making every user a cardiology patient.
This is the heart of the broader Healthcare Discovery question: how continuous measurement becomes useful care rather than just more data.
What the FDA List Reveals
The FDA’s cardiovascular entries show several major uses of AI and advanced software in heart care.
One group centers on rhythm. Systems such as DeepRhythmAI, iRhythm’s ZEUS platform, AliveCor products, VitalRhythm, Cardiologs Holter Platform, and other ECG-oriented tools sit in the world of rhythm detection, analysis, and monitoring. The clinical need is obvious. Arrhythmias can be intermittent, consequential, and easy to miss if the window of observation is too short.
Another group centers on cardiac function. Tools such as Tempus ECG-Low EF, Anumana’s ECG-AI Low Ejection Fraction algorithm, Bunkerhill ECG-EF, and Eko’s low-ejection-fraction work point toward a deeper possibility: ordinary cardiac signals may contain clues about structural or functional disease before the diagnosis is obvious.
A third group centers on heart sounds and imaging. Eko’s transformer-based heart-sound analysis, eMurmur Heart AI, echocardiography tools, and HeartFlow’s coronary analysis show how AI can help interpret signals beyond the ECG. The heart is electrical, mechanical, acoustic, and vascular. Each layer can become data.
A fourth group sits closer to remote monitoring and consumer-adjacent alerts. Apple’s Hypertension Notification Feature, Fitbit’s Loss of Pulse Detection, Withings ECG App, Empatica’s platform, Abbott’s Assert-IQ, and wearable cardiac defibrillator technology point toward a world where cardiac risk can be watched over longer periods and in more ordinary settings.
None of this means the heart has become easy to understand. It means cardiology is accumulating more ways to notice when something may be wrong.
The Risk of Too Much Signal
More measurement is not automatically better medicine.
Cardiac data can create clarity, but it can also create noise. A false alarm can frighten someone. A missed signal can create false reassurance. A pattern may be clinically meaningful in one person and incidental in another. A wearable notification may prompt appropriate follow-up, or it may send someone into a maze of anxiety and testing.
This is why FDA authorization matters, but also why it is not the end of the story. Authorization means a device or software function met the applicable regulatory requirements for a defined intended use. It does not mean the tool is perfect. It does not mean the function applies to every person in every context. It does not mean a consumer device has become a universal heart doctor.
Cardiology is full of thresholds, context, and judgment. A rhythm strip is interpreted differently depending on symptoms, age, medications, medical history, and risk. Blood pressure has meaning over time, not only in a single moment. A low ejection fraction signal may open the door to further evaluation, not close the case.
The best cardiac AI will not merely detect more. It will help separate what matters from what does not.
Why This Category Matters for Longevity
Cardiovascular disease remains one of the central problems in longevity because the heart and vascular system determine so much of healthspan. A person can feel well while blood pressure rises, plaque develops, rhythm changes, or early dysfunction begins. The first symptom can arrive late.
That makes cardiology different from many consumer wellness categories. A sleep score may help someone improve recovery. A step count may encourage movement. But a heart rhythm notification, an ECG algorithm, a blood-pressure pattern, or a cardiac function signal can point toward disease that carries immediate clinical stakes.
The opportunity is early detection without panic. More continuous cardiac measurement could help people catch atrial fibrillation sooner, recognize hypertension patterns earlier, identify deteriorating heart function before symptoms become severe, and connect the right signal to the right clinical follow-up.
The danger is turning every signal into a diagnosis.
That balance will define the next stage of heart monitoring. The heart is becoming more visible. The question is whether the new visibility leads to better decisions.
The Heart as the Bridge
Radiology became the first large beachhead for regulated medical AI because imaging is digital, structured, and central to diagnosis. Cardiology may become the bridge between clinical AI and everyday health because the heart can be measured in the clinic, at home, and on the body.
That bridge is already taking shape. It runs from hospital ECG systems to smartwatches, from echocardiography software to connected stethoscopes, from cardiac patches to remote monitoring dashboards, from coronary models to AI-derived risk signals.
The future of cardiac AI will not be one device. It will be a layered system of measurement, notification, interpretation, and follow-up. Some layers will be medical. Some will remain wellness. Some will live in hospitals. Some will live on wrists, chests, fingers, and phones.
The heart is teaching medicine what continuous AI-assisted monitoring may look like: not a single answer, but a stream of signals that needs intelligence, restraint, and clinical judgment.
Sources
- FDA: Artificial Intelligence-Enabled Medical Devices
- FDA: Examples of Device Software Functions the FDA Regulates
- Healthcare Discovery: FDA AI Medical Devices and the Quiet Reality of Medical AI